October 11, 2022
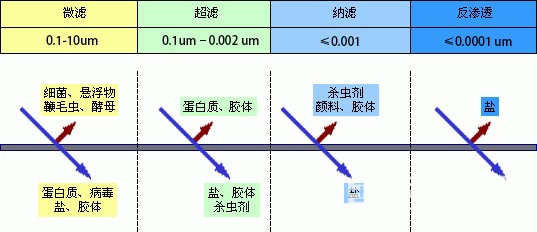
Water filtration technology can be divided into four types according to its fine degree, namely: microfiltration, ultrafiltration, nanofiltration and reverse osmosis, each filtration method treats different objects, and their filtration coarseness is also different, arranged by coarseness: microfiltration < ultrafiltration < nanofiltration < reverse osmosis, of course, the finer its filtration precision, the higher the purity of the clean processed.
The following is a brief discussion of these filtration methods
THE FIRST ONE
Microfiltration Concept
Microfiltration, also known as microporous filtration, is a separation process in which a porous membrane (microporous filter membrane) is used as the filtering medium to retain particles such as gravel, silt, clay and Giardia, Cryptococcus, algae and some bacteria in the solution under the pressure push of 0.1~0.3MPa, while a large number of solvents, small molecules and a small amount of macromolecular solutes are able to pass through the membrane.
Filtration accuracy: 0.1-10μm
Treatment objects:
Gravel, silt, clay and other particles, Giardia, Cryptococcus, algae and some bacteria, etc. Filtration principle.
Principle
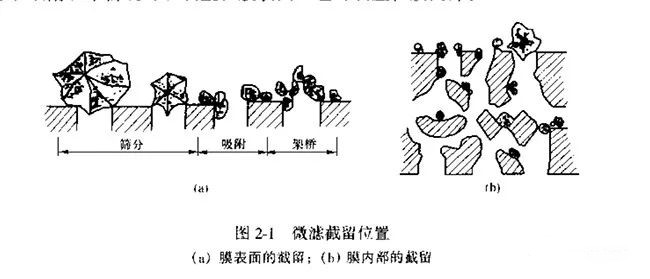
There are three types of filtration principles in microfiltration: sieving, cake layer filtration, and deep filtration. It is generally believed that the separation mechanism of microfiltration is sieving mechanism, and the physical structure of the membrane plays a decisive role. In addition, factors such as adsorption and electrical properties have an impact on the retention rate. The effective separation range is 0.1-10μm particles and the operating static pressure difference is 0.01-0.2MPa. according to the retention position of particles in the microfiltration process, it can be divided into three retention mechanisms: sieving, adsorption and bridging, and their microfiltration principles are as follows.
(1) Sieving: The microporous membrane intercepts particles that are larger than or comparable to the membrane pore size, also known as mechanical retention.
(2) Adsorption: Particles are adsorbed by the membrane through physical and chemical adsorption. Particle size smaller than the membrane pore can also be retained.
(3) Bridging: The particles accumulate and push each other, so that many particles cannot enter the membrane pore or get stuck in the pore, thus completing the retention.
Applications: pre-filter, ceramic water purifier, microporous membrane filter, security filter, etc.
THE SECOND ONE
Ultrafiltration Concept
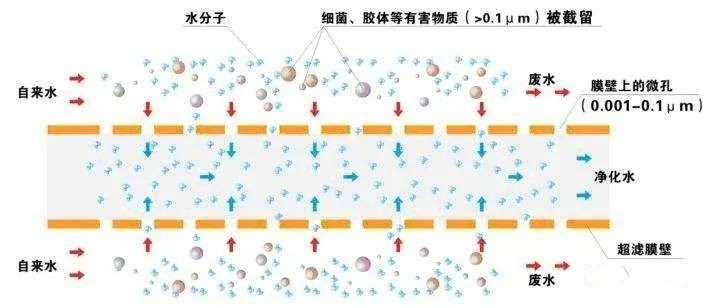
Ultrafiltration is the use of hollow fiber filtration new technology, with three levels of pretreatment filtration to remove impurities in tap water; ultrafiltration microporous less than 0.01 microns, can thoroughly filter out bacteria, rust, colloids and other harmful substances in the water, retaining the original trace elements and minerals in the water.
Filtration precision: 0.1um-0.002 um
Treatment objects:
Bacteria, rust, colloid filtration principle.
Principle
Ultrafiltration is a pressurized membrane separation technology, that is, under a certain pressure, small molecules of solutes and solvents pass through a special membrane of a certain pore size, while large molecules of solutes cannot pass through and remain on one side of the membrane, so that the large molecules are partially purified.
Ultrafiltration principle is also a membrane separation process principle, ultrafiltration using a pressure-activated membrane, under the action of external driving force (pressure) to retain water colloids, particles and relatively high molecular weight substances, while water and small solute particles through the membrane separation process.
Through the membrane surface microporous screening can retain the molecular weight of 3×10000-1×10000 substances.
When the treated water passes through the membrane surface at a certain flow rate with the help of external pressure, water molecules and solutes with molecular weight less than 300-500 pass through the membrane, while particles and macromolecules larger than the membrane pores are retained due to sieving, thus purifying the water.
In other words, when the water passes through the ultrafiltration membrane, most of the colloidal silica contained in the water can be removed, while a large amount of organic matter, etc. can be removed.
Applications: ultrafiltration water purifiers, ultrafiltration system, energy water machines, etc
THE THIRED ONE
Nanofiltration Concept
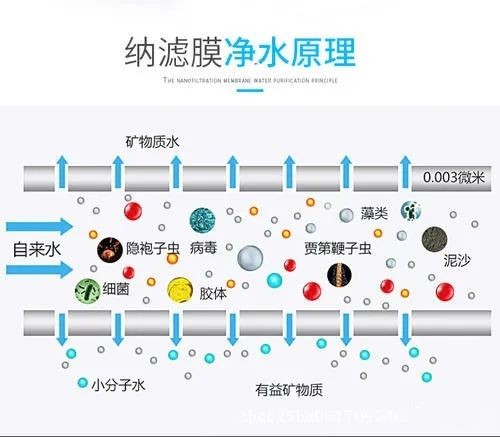
Filtration accuracy between ultrafiltration and reverse osmosis, can retain nanometer-level (0.001 micron) substances, desalination rate is lower than reverse osmosis, is a membrane separation technology that needs to be charged and pressurized, nanofiltration membrane is enough to retain substances with molecular weight of a few hundred, and the retention rate of certain low-molecular organic substances can reach 90%.
Since nanofiltration is more efficient in removing natural organic matter from water, it can also retain inorganic components of low molecular weight appropriately
Filtration precision: ≤0.001
Treatment object:
Organic matter as well as multivalent ions filtration principle.
Principle
Nanofiltration membranes are charged membranes and are capable of electrical adsorption. Under the same water quality and environment, the pressure required for nanofiltration membrane is less than the pressure required for reverse osmosis membrane. Therefore, in terms of separation principle, nanofiltration and reverse osmosis have similar and different aspects.
The pore size and surface characteristics of nanofiltration membrane determine its unique performance, and it has different Donann potential for ions with different charges and different valence numbers; the separation mechanism of nanofiltration membrane is sieving and dissolution diffusion, and it also has charge rejection effect, which can effectively remove divalent and multivalent ions, remove various substances with molecular weight greater than 200, and partially remove monovalent ions and substances with molecular weight less than 200.
The separation performance of nanofiltration membrane is significantly better than ultrafiltration and microfiltration, and compared with reverse osmosis membrane, it has the advantages of partial removal of monovalent ions, low process osmotic pressure, low operating pressure and energy saving
Application: Nanofiltration system, home nanofiltration water system, etc.
The FOURTHE ONE
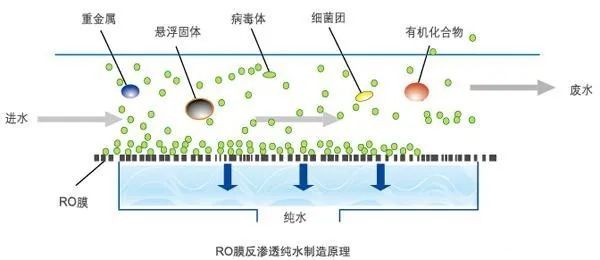
Reverse Osmosis Concept
Reverse osmosis, also known as reverse osmosis, is a membrane separation operation in which a pressure difference is used as a driving force to separate a solvent from a solution. It is called reverse osmosis because it is in the opposite direction to natural osmosis.
Depending on the different osmotic pressures of various materials, a reverse osmosis pressure greater than the osmotic pressure, i.e., reverse osmosis, can be used to achieve separation, extraction, purification and concentration.
Filtration accuracy: 0.0001 um
Treatment object:
Dissolved salts, colloids, microorganisms, organic matter, etc. in water Filtration principle.
Principle
The membrane that is selective for the permeable material is called a semi-permeable membrane, and the membrane that can only permeate the solvent but not the solute is generally called an ideal semi-permeable membrane.
When the same volume of dilute solution (such as fresh water) and concentrated solution (such as salt water) are placed on both sides of the semi-permeable membrane, the solvent in the dilute solution will naturally pass through the semi-permeable membrane and flow spontaneously to the concentrated solution side, a phenomenon called osmosis.
When osmosis reaches equilibrium, the liquid level on the concentrated solution side will be higher than that of the dilute solution by a certain height, i.e., a pressure difference is formed, and this pressure difference is the osmotic pressure.
The magnitude of osmotic pressure depends on the intrinsic nature of the solution, i.e., it is related to the type, concentration and temperature of the concentrated solution and not to the nature of the semipermeable membrane.
If a pressure greater than the osmotic pressure is applied on the side of the concentrated solution, the flow direction of the solvent will be opposite to the original direction of osmosis and will begin to flow from the concentrated solution to the side of the dilute solution, a process called reverse osmosis.
Reverse osmosis is a reverse migration movement of osmosis, which is a pressure-driven separation method that separates solutes and solvents in solution with the help of selective retention of semi-permeable membranes, and it has been widely used in the purification and concentration of various liquids, the most common application example is in the water treatment process, using reverse osmosis technology to remove inorganic ions, bacteria, viruses, organic matter and colloids and other impurities from raw water to Obtain high quality pure water
Applications.
Reverse osmosis water purification system, home water purifier system, commercial RO system, direct drinking water system, etc
These are the 4 common water filtration methods, each filtration method for different occasions, play a different role, they are not good or bad, only according to their own needs, choose the right filtration method!